Physicists model cell migration to learn how cancer cells navigate tissue
Their model could bring scientists closer to understanding — and predicting — how cancer cells move as they invade the body during metastasis.
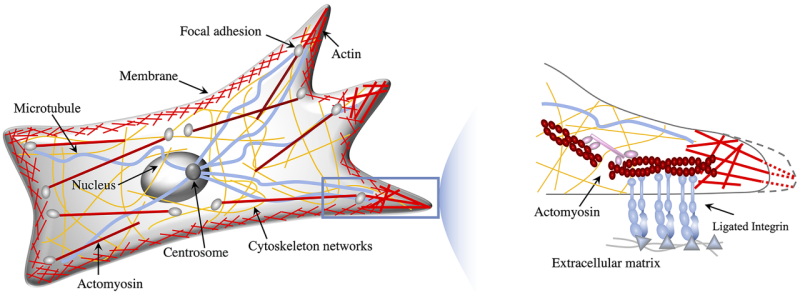
During mesenchymal migration, a cancer cell moves like a gecko on a wall.
Before choosing this mode of movement, the cell sizes up the surface to which it may stick, explained physicist Nadir Kaplan. If the surface isn’t too stiff or soft and the path forward isn’t too constricting, the round cell will rapidly grow protrusions that act like temporary limbs, jutting forward and sticking to the surface. The cell will then pull itself forward and retract its rear and repeat the process.
This migration mode is one of the ways cancer cells navigate tissue during metastasis. In a study published by Biophysical Journal, postdoctoral physics fellow Wenya Shu and Kaplan, an assistant professor of physics in the College of Science, explored mesenchymal migration through cell simulations and mathematical modeling. Their aim: to learn more about how cancer cells size up surrounding tissue for stiffness and adapt their gecko-like movements in response.
The model and its insights on mesenchymal migration are a first step in learning how cancer cells migrate on the whole, Shu said. Cell migration is complex: Cells make use of multiple modes of migration, both individually and in colonies. “That’s the advantage of coming up with a computational model here,” he said. “We can dissect the effects of many ingredients at play.”
Experiments show that during mesenchymal migration, cells adapt how they navigate tissue based on stiffness: They’re drawn to tissue surfaces — or substrates — that are not too stiff or too soft. Cells can’t grow and attach their protrusions efficiently to too-stiff substrates, and if cells grab onto too-soft tissue, they’ll end up pulling it back toward their bodies, rather than using it to pull themselves forward. Shu and Kaplan’s cell simulations backed these experimental findings.
Their simulations confirmed to the researchers that cells distinguish between soft and stiff surfaces by comparing them to the physical properties of their own soft bodies. Substrate material properties will then affect the directions cells take as well as how efficiently they move.
To ensure that the model accurately simulated cancer cell migration, Kaplan and Shu built in not only how cells respond to the substrate mechanics of tissue, but also how they tune their internal biochemical signals. While navigating tissue, cells may also respond chemically to the secretions of a nutrition source in the body. The researchers’ model is the first to simulate how both of these drivers of cell motion play out, Shu said.
The researchers found that cells prefer moving in the direction determined by their strong internal chemical signaling, whether or not the overall motion is efficient. But without a strong chemical signal to follow, they focus on substrate properties.
By piecing these elements of mesenchymal migration together and reproducing them in a model, Kaplan sees a move toward better understanding and pinpointing how and where metastasis may occur.
Metastasis also may involve multiple cell migration modes. Mesenchymal migration tends to be the initial mode of migration through tissue and into vessels, but cells often pivot to amoeboid migration. Whereas cells move like geckos in the former mode, the latter has them moving more like tank treads. “They just roll forward,” Kaplan said.
Chemotherapy works well against cancer cells in mesenchymal migration, Kaplan said, but not as well when the cells switch to amoeboid migration. For experimentalists to understand that transition, they first need a better grasp of the mesenchymal mode.
That is what we have made progress toward here,” Kaplan said.
Next, Shu and Kaplan hope to use the model to look at how cell-cell interactions may affect migration, as individual cells bump into one another and set off changes in their direction. They also want to learn how cells negotiate more curved, narrow channels in their microenvironment.
Each effort to more closely model cell migration brings the team closer to understanding how cancer cells invade the body. “We want to come up with a predictive model that can produce new types of qualitative behaviors, to explain more measurements and motivate new experiments,” Kaplan said.
“Experiments are quite comprehensive, but they significantly benefit from simulations. For instance, when it comes to resolving very small time scales in the dynamics of these cell deformations. We are basically discerning all those components,” he said.






