Hopping the species barrier: Harmful bacteria discovered in both amphibians and mammals
Brucella bacteria have a not-so-illustrious reputation for causing illness and death in mammals.
In the U.S. alone, Brucella infections cause economic losses on the order of $30 million per year due to dead livestock, infected milk, and general health care costs. In countries that rely heavily on agricultural products, it presents an even greater threat.
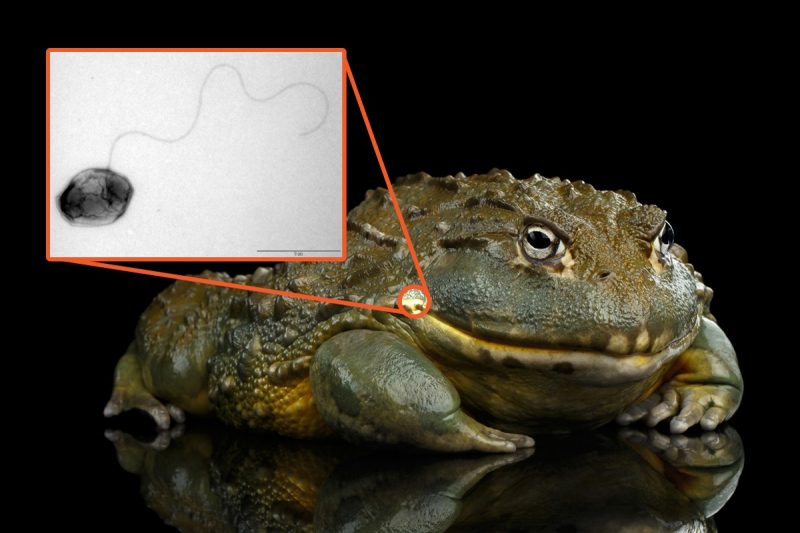
Once thought to affect mainly humans and livestock, Brucella is now being found in species scientists never expected. Previously unknown strains of the bacteria were recently discovered in frogs.
Scientists in the Biocomplexity Institute of Virginia Tech’s PATRIC group are examining genetic sequences of Brucella from these amphibian isolates and comparing them to more well-known mammalian strains.
Their findings, recently published in Scientific Reports, revealed that amphibian Brucella strains show distinctive characteristics not seen in other forms — the most surprising development being a whip-like flagellum that helps the bacteria move through their environment. These discoveries are radically changing how we think of disease and the transfer of bacteria between organisms.
“Since the 1850s, we had the idea of Brucella bacteria as having strong preferences for particular hosts,” said Rebecca Wattam, research assistant professor at the Biocomplexity Institute. “Now, we know that these bacteria are much more diverse than we’d previously thought. Our view of the Brucella world is expanding dramatically.”
The strains of Brucella found in African bullfrogs were cultured and found to be able to live in mammalian hosts for up to 12 weeks, suggesting that new types of brucellosis infections may be headed our way. In fact, there have been two recent cases where previously unseen strains of Brucella were found in a human host.
Though these discoveries present significant cause for concern, new strains of Brucella and other pathogens might help scientists to better understand how microorganisms evolve and transfer their genetic material.
Metagenomic analysis makes finding these new strains of bacteria possible. Instead of culturing strains in a lab and sequencing their genomes, scientists are able to extract samples for analysis from entirely new sources. The ability to isolate DNA from sources like soil is likely to speed up the discovery of new organisms, providing us with what might be our first real glimpse into the diversity of microbiota.
“It’s a great time to be a biologist, especially with metagenomic analysis,” Wattam said. “We know some things really well, but we now have the tools to explore what we never imagined. In light of all this new information, we definitely need new ways of looking at bacteria and how we live with them.”